About the Webinar
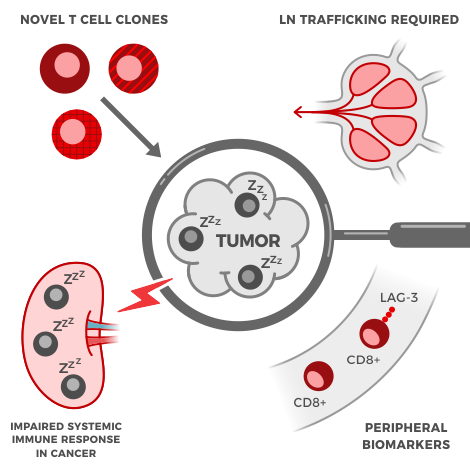
About ~40-60%[1] of first-line metastatic melanoma patients receive immunotherapy. But depending on the type of treatment, between 15-50% of those patients will have a severe immune-related adverse event of grade 3 or higher. These aren’t headaches either; the adverse events are serious, like Grade 3 hepatitis or colitis. These kinds of events “interfere with a person’s ability to do basic things like eat or get dressed.”[2] The problem is, drug developers and doctors don’t know which kind of patients will experience these adverse events. That’s why we’re attempting to crack this problem, by looking at immune signatures found in the blood of melanoma patients.
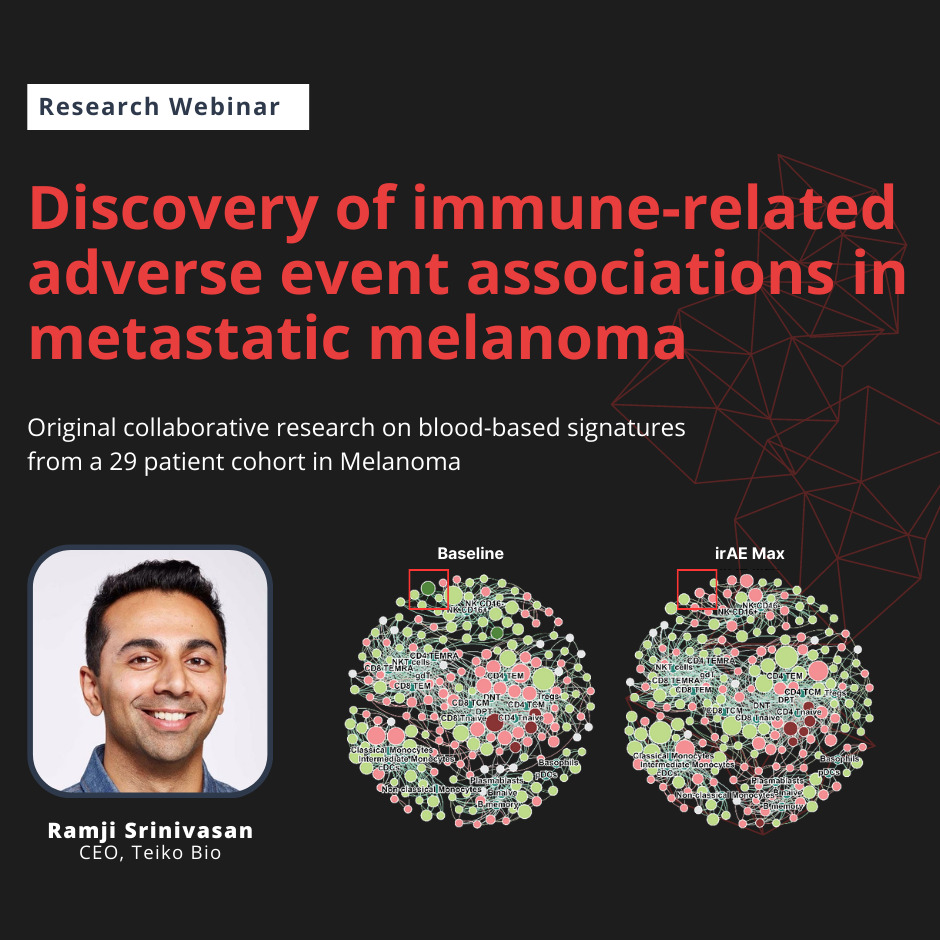
In this recorded live webinar, Ramji Srinivasan, cofounder and CEO of Teiko Bio, shares original research on blood-based biomarkers of immune-related adverse events in metastatic melanoma performed by Teiko scientists in collaboration with Dr. Siwen Hu-Lieskovan of Huntsman Cancer Institute.
About the Speaker
Before Teiko, Ramji was Cofounder, CEO and Chairman of Counsyl, a women’s health genetic screening laboratory. Counsyl screened over 1M prospective parents, mothers-to-be and women at risk of hereditary cancer. In 2018, Counsyl was acquired by Myriad Genetics, Inc for $375M in cash and stock.
Ramji earned a B.S. in computer science and an M.S. in financial mathematics, both from Stanford University. Ramji also attended Stanford’s Graduate School of Business before dropping out to start Counsyl.